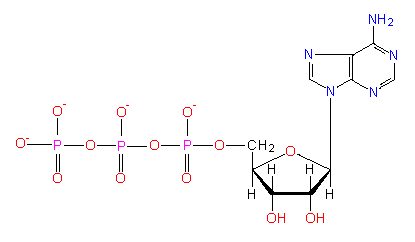
The ATP molecule
ATP is regarded as a universal source of energy in all kinds of cells. It is produced mainly in the oxidizing of energy-rich (reduced) compounds in the course of the respiratory chain and in photosynthesis. From Enzymes are life's catalysts:: Adenosine triphosphate is the primary molecule to which energy is transferred during the breakdown of fuel molecules, carbohydrates and fats. (A cell cannot use heat energy to perform its functions). ATP is then hydrolyzed to release energy which can be used by the energy requiring processes in cells such as (1) production of force and movement, (2) active transport across membranes and (2) synthesis of organic molecules used in cell structures and functions. ATP is an energy transfer molecule and NOT an energy storage molecule. It transfers energy from fuel molecules to cells in small amounts OK, so energy is being transferred - but what does this really mean? What exactly is being transported around and how does the body take advantage of this? I've found very interesting answers: 1.) When energy (such as in a photon) is pumped into a chemical system, the energy partitions into thermal and electronic components. The thermal component makes the molecules move faster, and the electronic component increases the number of "high-energy" electronic states. Both energy components will foster molecular organization: the faster the molecules vibrate, rotate, and translate, and the more of them that are in electronic states above ground level, the higher is the probability that the molecules will interact and the more work can be done in organizing them. ...
Living organisms store photon energy in chemical form, and then trickle it down molecular chains to the individual molecular bonding sites. ...
The photon energy is stored in the covalent bonds of glucose -- about 6 quanta of photon in one glucose molecules. From this reservoir, energy then flows along various pathways, nursing everything, all organization and all work. The chemical energy chains that nurse macromolecular organization commonly use ATP as their final link. (ref.) 2.) From Trudy Wassenaar: Many different proteins extract energy from the hydrolysis of ATP. All of them appear to couple some sort of motion of the protein with the energy-releasing hydrolysis. The motion of the protein can in turn be harnessed to do necessary things, such as pump chemicals across a cell membrane, move the cell around, synthesize new proteins, and so on. 3.) Remember the Ion Pump description that won Jens C. Skou the 1997 Nobel prize? Well, he shared the 1997 Nobel Prize in Chemistry with Paul D. Boyer, John E. Walker "for their elucidation of the enzymatic mechanism underlying the synthesis of adenosine triphosphate (ATP). Check out Boyer's lecture "Energy, Life and ATP", and Walker's lecture"ATP Synthesis by Rotary Catalysis". Digging further back in time brings you to the 1953 The Nobel Prize in Physiology or Medicine awarded to Hans Krebs for his work on the Citric Acid Cycle, and Fritz Lipmann for his discovery of "co-enzyme A and its importance for intermediary metabolism". Lipmann talks about "energy-rich phosphate bonds" and "transformations of electron transfer potential ... to phosphate bond energy". John Kyrk's put together two very cool animations that show how the Krebs citric acid cycle works and how mitochondria create ATP.
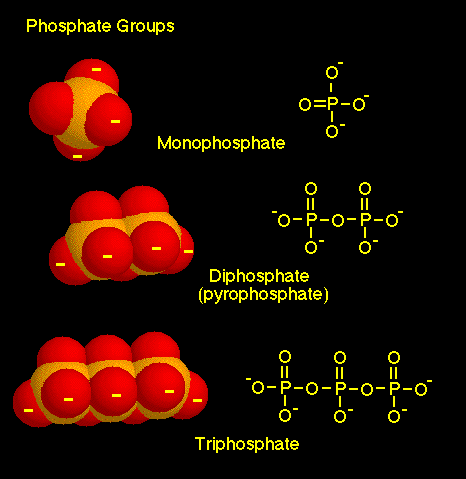 4.) From University of Connecticut: When phosphate groups are joined together, they have a strong tendency to repel each other, because of the high concentration of negative charge in the very polar and usually ionized oxygen atoms. [the O- in the ATP molecular diagram] As a result, molecules with two or three phosphate groups are good energy donors, readily releasing energy along with the transfer of phosphate groups.
4.) From University of Connecticut: When phosphate groups are joined together, they have a strong tendency to repel each other, because of the high concentration of negative charge in the very polar and usually ionized oxygen atoms. [the O- in the ATP molecular diagram] As a result, molecules with two or three phosphate groups are good energy donors, readily releasing energy along with the transfer of phosphate groups.
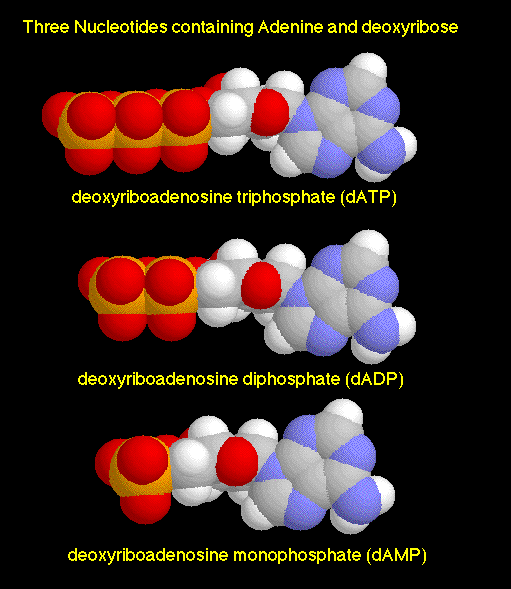 It feels like a number of things that have been discussed in these notes are finally starting to come together. Where else have we seen these phosphate groups before? Oh yeah - the nucleotides that make up DNA. It's not hard to imagine the ionized O- atoms associated with all of those phosphate groups kicking the molecules around energetically, making sure that they encounter the other molecules that they need to form bonds and ligands with. And why don't these O- atoms bond with, say Na+ or K+ ions? For the same reason that we don't find too many NaCl crystals in the body - they are dissolved by water.
More images and text from the great webpage at the University of Hamburg:ATP and Other Nucleoside Triphosphates or: Links Rich in Energy
It feels like a number of things that have been discussed in these notes are finally starting to come together. Where else have we seen these phosphate groups before? Oh yeah - the nucleotides that make up DNA. It's not hard to imagine the ionized O- atoms associated with all of those phosphate groups kicking the molecules around energetically, making sure that they encounter the other molecules that they need to form bonds and ligands with. And why don't these O- atoms bond with, say Na+ or K+ ions? For the same reason that we don't find too many NaCl crystals in the body - they are dissolved by water.
More images and text from the great webpage at the University of Hamburg:ATP and Other Nucleoside Triphosphates or: Links Rich in Energy
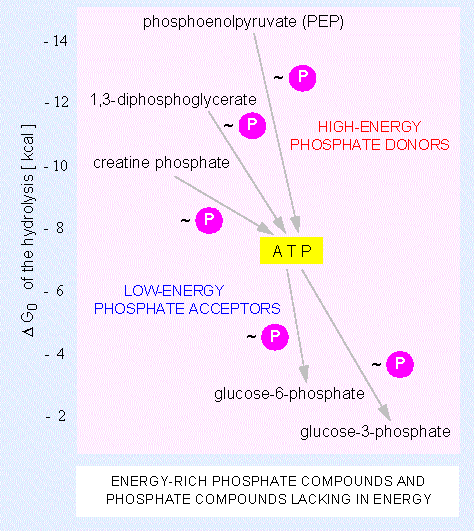
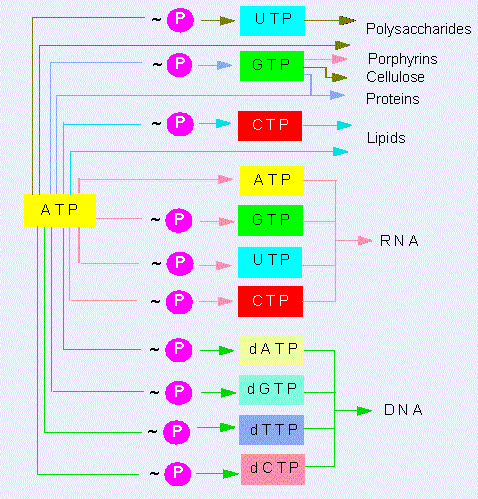
Besides the adenosine nucleotide phosphates occur also uracil, cytosine and guanine phosphates: UMP, UDP, UTP, CMP, CDP, CTP, GMP, GDP, GTP. The triphosphate nucleosides of the mentioned compounds including ATP are components of RNA. They are integrated into the polymer by cleavage of pyrophosphate ( = PP). The corresponding desoxyribose derivatives (dATP, dGTP, dCTP....) are necessary for DNA synthesis where dTTP is used instead of dUTP. The terminal phosphate residues of all nucleoside di- and triphosphates are equally rich in energy. The energy set free by their hydrolysis is used for biosyntheses. They share the work equally: UTP is needed for the synthesis of polysaccharides, CTP for that of lipids and GTP for the synthesis of proteins and other molecules. These specificities are the results of the different selectivities of the enzymes that control each of these metabolic pathways.
No comments:
Post a Comment