
From Stuart Kauffman's essay "What is Life?" in the book "The next fifty years": An enclosed gas in a thermodynamically isolated box can do no work. But if the box is divided into two parts by a membrane, then one part can do work on the other part; for example, if the gas pressure is higher in the first part, the membrane will bulge into the second part, doing mechanical work on it. Thus work cannot be achieved in the universe unless the universe is divided into at least two regions. Furthermore, just where did the membrane come from?
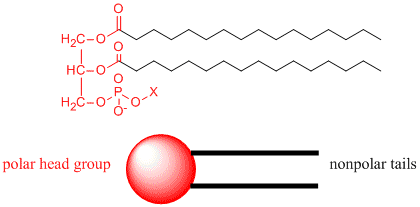 For cells, the membrane is made up of molecules that are polarized on one end - i.e. their atoms are physically arranged so that another molecule can get close enough to the molecule to feel the inter molecular force exerted by the presence or lack of electrons around one atom - and unpolarized at the other end. The polarized ends are 'hydro-philic' and are attracted to water, whereas the non-polar ends are hydrophobic and get pushed away from the water molecules.
For cells, the membrane is made up of molecules that are polarized on one end - i.e. their atoms are physically arranged so that another molecule can get close enough to the molecule to feel the inter molecular force exerted by the presence or lack of electrons around one atom - and unpolarized at the other end. The polarized ends are 'hydro-philic' and are attracted to water, whereas the non-polar ends are hydrophobic and get pushed away from the water molecules. 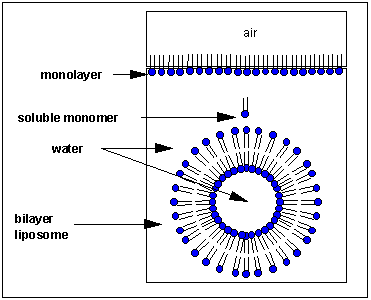
From Henry Jakubowski's page at St. John's University: How would you orient this molecule in water? There are several possible ways. A small number of these molecules might be soluble in water as above. Mostly, however, the nonpolar tails wants to get out of the water, while the polar head like to stay in the water. Again, some of the molecules migrate to the surface of the water, with the nonpolar tails sticking out into air, away from water, to form a monolayer on the top of the water. Others will self- aggregate, through Inter-Molecular Forces (IMF's) to form a bilayer or membrane. Because there are two tails per head group, the tails can't pack together as tightly. Imagine the bilayer or membrane curving around and eventually meeting. A structure like this would look like a small biological cell. ... The interior of this little cell, or liposome, is filled with water.
Now, cell membranes are not rigid or impermeable. In fact, things like proteins with one hydrophobic end can push into them and get embedded in them fairly easily. Ions (like Na+ or K+, for example) are not able to pass through the membrane. Also, the acidity of the water inside the liposome can become different than outside of the liposome. This sets up the conditions for molecules to diffuse through the membrane, as ions are driven by the concentration gradient to spread to regions of lower concentration. i.e. it sets up the conditions needed to get some work done!
This still leaves Stuart Kauffman's question - "Just where did the membrane come from?"
From First Cell by Carl Zimmer: Life is chemical interaction, and for that interaction to occur, life’s molecules must be close to one another. Without a physical boundary of some sort, without a skin, a bark, or a cell membrane, an organism is nothing more than a diffusing blur of molecules. To explain how the first creature came to be, you have to explain how its innards got to be distinguished from its surroundings. In other words, you’ve got to explain how the first single- celled creature got encapsulated in a cell.
...
A cell membrane’s importance to life is often underappreciated, says David Deamer (University of California at Santa Cruz). People say, ‘Well, it’s just a little bag.’ But it’s much more. It’s the interface between life and everything that’s outside. The membrane of any cell has to do many things at once. It has to be impermeable enough to keep essential things (like DNA) in and harmful things (like viruses and poisons) out. Yet a cell membrane can’t form a perfect seal. It has to be able to flush out waste and heat from its own system and take in nutrients from the surrounding medium. And the first cell membrane, like the membranes of many single-celled organisms today, probably had to be able to collect energy as well. No sense repeating the whole article here - it's a fascinating read on how life might have gotten started - and membranes look like they were the key.
1 comment:
Yes, membranes are where the action is!! Read Bruce Lipton's "The Biology of Belief" in which he talks about the brain of the cell actually being the membrane NOT the nucleus. It changes one's perspective in cellular biology.
Post a Comment