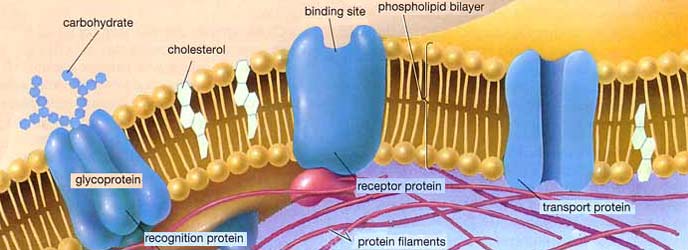
 Cell membranes are around 7nm thick, and the lipid bi-layers that they are made of are highly impermeable to ions. The membranes are studded with numerous proteins that have a wide variety of functions. Some of these proteins, for example, form pores that allow molecules to flow from one side of the cell membrane to the other. Hydrophobic amino acids line the outside of the pore, binding the pore into the membrane, and hydrophilic amino acids line the inside of the pore, providing a channel that is friendly to water-soluble chemicals. (ref.)
The characteristics of the pore are determined by the nature of the protein molecule it is made of. For example, if it is made of a protein called aquaporin, it will create a hydrophilic channel through the cell membrane that has a passage big enough for water molecules to pass through but too small for substances such as alcohol. Certain types of pore molecules are 'gated' so that they can be shut or opened in response to certain conditions. Pores that allow specific ions to pass through the cell membrane are called 'ion channels'.
(more info on cell membranes and pores)
From HHMI - "Visualizing a Potassium Channel": Around 50 years ago researchers showed that electrical activity in neurons is produced by subtle changes in the neuron's potassium concentration. "Since then, it's been well established that the flow of potassium ions is central to many different cellular processes," said MacKinnon. Potassium currents in the brain, for example, underlie perception and movement, and the heart's contraction relies upon the steady ebb-and-flow of potassium.
To maintain the correct concentration of potassium, cells are equipped with pore-like proteins that poke through the cell membrane. These proteins, called ion channels or potassium channels, create sieves through which potassium ions flow from inside to outside the cell.
Channels are just one mechanism to move ions in and out of the cell, however. There are also passive carriers (molecules that enclose ions and move by following chemical or electrical gradients), and active carriers such as ion pumps. The main difference between channels and carriers is that with carrier proteins there is never an open channel all the way through the membrane. (ref.)
Ion Channels
Cell membranes are around 7nm thick, and the lipid bi-layers that they are made of are highly impermeable to ions. The membranes are studded with numerous proteins that have a wide variety of functions. Some of these proteins, for example, form pores that allow molecules to flow from one side of the cell membrane to the other. Hydrophobic amino acids line the outside of the pore, binding the pore into the membrane, and hydrophilic amino acids line the inside of the pore, providing a channel that is friendly to water-soluble chemicals. (ref.)
The characteristics of the pore are determined by the nature of the protein molecule it is made of. For example, if it is made of a protein called aquaporin, it will create a hydrophilic channel through the cell membrane that has a passage big enough for water molecules to pass through but too small for substances such as alcohol. Certain types of pore molecules are 'gated' so that they can be shut or opened in response to certain conditions. Pores that allow specific ions to pass through the cell membrane are called 'ion channels'.
(more info on cell membranes and pores)
From HHMI - "Visualizing a Potassium Channel": Around 50 years ago researchers showed that electrical activity in neurons is produced by subtle changes in the neuron's potassium concentration. "Since then, it's been well established that the flow of potassium ions is central to many different cellular processes," said MacKinnon. Potassium currents in the brain, for example, underlie perception and movement, and the heart's contraction relies upon the steady ebb-and-flow of potassium.
To maintain the correct concentration of potassium, cells are equipped with pore-like proteins that poke through the cell membrane. These proteins, called ion channels or potassium channels, create sieves through which potassium ions flow from inside to outside the cell.
Channels are just one mechanism to move ions in and out of the cell, however. There are also passive carriers (molecules that enclose ions and move by following chemical or electrical gradients), and active carriers such as ion pumps. The main difference between channels and carriers is that with carrier proteins there is never an open channel all the way through the membrane. (ref.)
Ion Channels Ion channels have to be able to admit one ion type selectively, but not another, and to open and shut and sometimes to conduct ions in one direction only. But until recently, it was not known how this molecular machinery really worked.
From U. of Miami - Ion channels: During the 1970s it was shown that the ion channels were able to admit only certain ions because they were equipped with some kind of "ion filter". Of particular interest was the finding of channels that admit potassium ions but not sodium ions – even though the sodium ion is smaller than the potassium ion. It was suspected that oxygen atoms in the transport protein played an important role as "substitutes" for the water molecules with which the potassium ion surrounds itself in a solution of water and from which it must free itself during entry to the channel. Only the Ion can pass, not it hydrated counterpart.
In 1998 Roderick MacKinnon determined the first high-resolution structure of an ion channel and revealed for the first time how an ion channel functions at atomic level... it uses an ion filter, which admits potassium ions and stops sodium ions. His ability with X-ray crystallography made it possible to unravel how the ions passed through the channel. The ions could also be visualized in the crystal structure – surrounded by water molecules just before they enter the ion filter; right in the filter, and when they meet the water on the other side of the filter.
Ion channels have to be able to admit one ion type selectively, but not another, and to open and shut and sometimes to conduct ions in one direction only. But until recently, it was not known how this molecular machinery really worked.
From U. of Miami - Ion channels: During the 1970s it was shown that the ion channels were able to admit only certain ions because they were equipped with some kind of "ion filter". Of particular interest was the finding of channels that admit potassium ions but not sodium ions – even though the sodium ion is smaller than the potassium ion. It was suspected that oxygen atoms in the transport protein played an important role as "substitutes" for the water molecules with which the potassium ion surrounds itself in a solution of water and from which it must free itself during entry to the channel. Only the Ion can pass, not it hydrated counterpart.
In 1998 Roderick MacKinnon determined the first high-resolution structure of an ion channel and revealed for the first time how an ion channel functions at atomic level... it uses an ion filter, which admits potassium ions and stops sodium ions. His ability with X-ray crystallography made it possible to unravel how the ions passed through the channel. The ions could also be visualized in the crystal structure – surrounded by water molecules just before they enter the ion filter; right in the filter, and when they meet the water on the other side of the filter.

OUTSIDE THE ION FILTER (upper fig.)
Outside the cell membrane the ions are bound to water molecules with certain distances to the oxygen atoms of the water. INSIDE THE ION FILTER (lower fig.)
For the potassium ions the distance to the oxygen atoms in the ion filter is the same as in water. The ion channel is able to strip the potassium ion of its water and allow it to pass at no cost in energy. The sodium ions, which are smaller, do not fit in between the oxygen atoms in the filter. This prevents them from entering the channel.
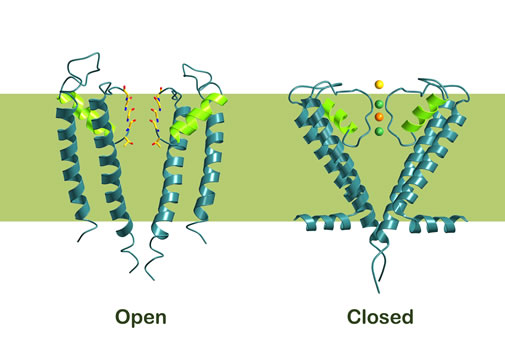 Cells must also be able to control the opening and closing of ion channels. MacKinnon has shown that this is achieved by a gate at the bottom of the channel which opened and closed a molecular “sensor”. This sensor is situated close to the gate. Certain sensors react to certain signals, e.g. an increase in the concentration of calcium ions [Ligand Gated Channel], an electric voltage over the cell membrane, [Voltage Gated Channel], or binding of a signal molecule of some kind [Stress Activated Gated Channel]. By evolving different molecular sensors to ion channels, nature has created ion channels that respond to a large number of different environmental signals.
Passive Ion Carriers
Cells must also be able to control the opening and closing of ion channels. MacKinnon has shown that this is achieved by a gate at the bottom of the channel which opened and closed a molecular “sensor”. This sensor is situated close to the gate. Certain sensors react to certain signals, e.g. an increase in the concentration of calcium ions [Ligand Gated Channel], an electric voltage over the cell membrane, [Voltage Gated Channel], or binding of a signal molecule of some kind [Stress Activated Gated Channel]. By evolving different molecular sensors to ion channels, nature has created ion channels that respond to a large number of different environmental signals.
Passive Ion CarriersFrom NIH - Molecular Biology of the Cell: Many carrier proteins allow solutes to cross the membrane only passively ("downhill"), a process called passive transport, or facilitated diffusion. In the case of transport of a single uncharged molecule, it is simply the difference in its concentration on the two sides of the membrane (its concentration gradient) that drives passive transport and determines its direction. Ionophores are small hydrophobic molecules that dissolve in lipid bilayers and increase their permeability to specific inorganic ions. ... [They] operate by shielding the charge of the transported ion so that it can penetrate the hydrophobic interior of the lipid bilayer. Since ionophores are not coupled to energy sources, they permit the net movement of ions only down their electrochemical gradients. Valinomycin is an example of a mobile ion carrier. It is a ring-shaped polymer that transports K+ down its electrochemical gradient by picking up K+ on one side of the membrane, diffusing across the bilayer, and releasing K+ on the other side.
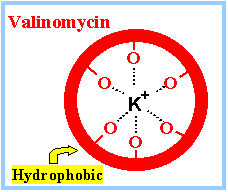
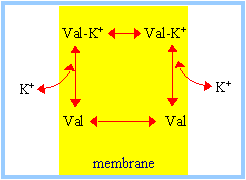
Similarly, FCCP, a mobile ion carrier that makes membranes selectively leaky to H+, is often used to dissipate the H+ electrochemical gradient across the mitochondrial inner membrane, thereby blocking mitochondrial ATP production. A23187 is yet another example of a mobile ion carrier, only it transports divalent cations such as Ca2+ and Mg2+. When cells are exposed to A23187, Ca2+ enters the cytosol from the extracellular fluid down a steep electrochemical gradient. Accordingly, this ionophore is widely used to increase the concentration of free Ca2+ in the cytosol, thereby mimicking certain cell-signaling mechanisms. (more...) Ion Pumps
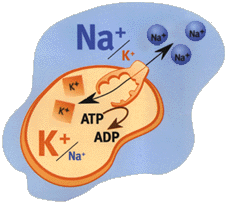 From NobelPrize.org - The first Ion pump discovered (1997 Nobel Prize):Ion composition outside the cell differs from that inside. When e.g. a nerve is stimulated Na+ flows into the cell. The electrical potential arising across the membrane causes the nerve impulse to propagate itself along the nerve. The normal state is restored when Na+ has been pumped out and K+ pumped in.
Jens Skou discovered Na+, K+-ATPase - an enzyme that maintains the balance of sodium (Na+) and potassium (K+) ions in cells. Within cells, the concentration of Na+ ions is lower, and that of K+ ions higher, than in the surrounding fluid.
Na+, K+-ATPase and other ion pumps must work all the time in our body. If they were to stop, our cells would swell up, and might even burst, and we would rapidly lose consciousness. A great deal of energy is needed to drive ion pumps - in humans, about 1/3 of the ATP that the body produces.
The Na+, K+-ATPase cycle:
From NobelPrize.org - The first Ion pump discovered (1997 Nobel Prize):Ion composition outside the cell differs from that inside. When e.g. a nerve is stimulated Na+ flows into the cell. The electrical potential arising across the membrane causes the nerve impulse to propagate itself along the nerve. The normal state is restored when Na+ has been pumped out and K+ pumped in.
Jens Skou discovered Na+, K+-ATPase - an enzyme that maintains the balance of sodium (Na+) and potassium (K+) ions in cells. Within cells, the concentration of Na+ ions is lower, and that of K+ ions higher, than in the surrounding fluid.
Na+, K+-ATPase and other ion pumps must work all the time in our body. If they were to stop, our cells would swell up, and might even burst, and we would rapidly lose consciousness. A great deal of energy is needed to drive ion pumps - in humans, about 1/3 of the ATP that the body produces.
The Na+, K+-ATPase cycle: 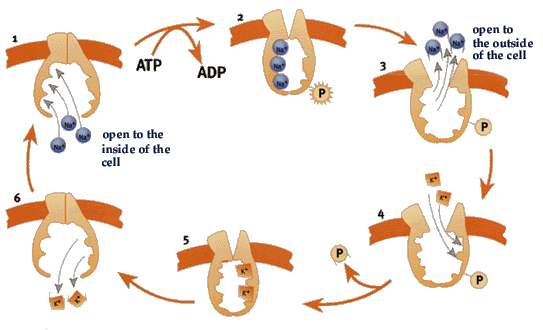 Fig 1.) The enzyme opens up towards the inside of the cell and exposes binding sites for three Na+ ions.
Fig 2.)When the ions are bound, an ATP molecule binds to the enzyme and one of its phosphate groups is transferred.
Fig. 3) The enzyme changes shape, opening towards the outside and the Na+ ions are released.
Fig. 4) Now binding sites for two K+ ions are exposed.
Fig 1.) The enzyme opens up towards the inside of the cell and exposes binding sites for three Na+ ions.
Fig 2.)When the ions are bound, an ATP molecule binds to the enzyme and one of its phosphate groups is transferred.
Fig. 3) The enzyme changes shape, opening towards the outside and the Na+ ions are released.
Fig. 4) Now binding sites for two K+ ions are exposed.
No comments:
Post a Comment